Revolutionizing Water Treatment: The Science Behind Catalytic Reactive Membranes
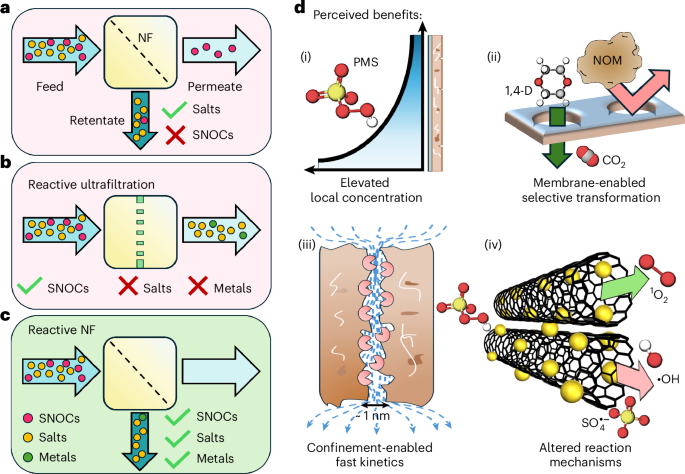
Reactive nanofiltration membranes are at the forefront of water treatment technology, combining catalytic transformation with molecular separation to tackle diverse aqueous contaminants. Despite their potential, the development of these membranes has been slowed by gaps in understanding the interaction between solute mass transport and chemical reactions.
Key Design Principles Unveiled
Through a systematic modeling approach, researchers have identified critical design principles that enhance membrane performance:
- Efficient Oxidant Transport: Essential for maximizing contaminant degradation.
- Catalyst Utilization: For surface-loaded catalysts, avoiding mass transport limitations is key. For interior-loaded catalysts, optimizing oxidant partitioning boosts efficiency.
- Selective Solute Rejection: Reduces interference from natural organic matter, enabling more selective contaminant transformation within membrane pores.
Performance Insights
- At low permeate water fluxes, surface-catalysed reactions dominate contaminant transformation.
- At high fluxes, interior-catalysed reactions take precedence.
- Strategic membrane design can minimize secondary contamination by rejecting salts produced during catalytic reactions.
Enhancing Treatment Performance
Selecting the right oxidant–catalyst pairs can generate suitable reactive species, further improving treatment efficiency. This study not only provides a theoretical framework for designing reactive nanofiltration membranes but also offers insights into the development of advanced water treatment technologies.




Comments
Join Our Community
Sign up to share your thoughts, engage with others, and become part of our growing community.
No comments yet
Be the first to share your thoughts and start the conversation!